Review Labs Diffusion and Osmosis
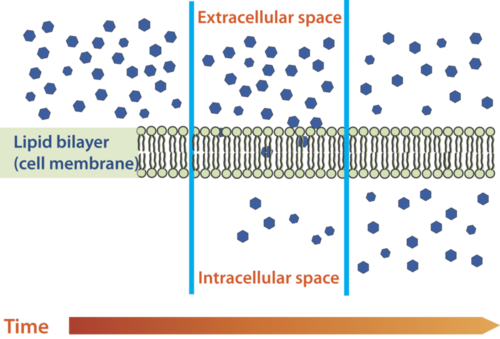
Diffusion is the movement of molecules from an area of high concentration of the molecules to an area with a lower concentration. The difference in the concentrations of the molecules in the two areas is called the concentration gradient. Diffusion will continue until this gradient has been eliminated. Since diffusion moves materials from an area of higher concentration to the lower, it is described as moving solutes "down the concentration gradient." The end result of diffusion is an equal concentration, or equilibrium, of molecules on both sides of the membrane.
Summary
Summary
Summary
- The cell membrane is selectively permeable, allowing only certain substances to pass through.
- Passive transport is a way that small molecules or ions move across the cell membrane without input of energy by the cell. The three main kinds of passive transport are diffusion, osmosis, and facilitated diffusion.
- Diffusion is the movement of molecules from an area of high concentration of the molecules to an area with a lower concentration.
Summary
Diffusion Illustration
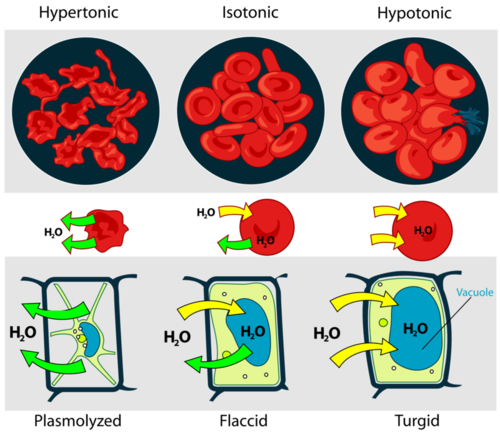
Osmosis Illustration
Review
1. What is diffusion?
2. What is a concentration gradient?
3. What is meant by passive transport?4. What is osmosis? What type of transport is it?
5. How does osmosis differ from diffusion?
1. What is diffusion?
2. What is a concentration gradient?
3. What is meant by passive transport?4. What is osmosis? What type of transport is it?
5. How does osmosis differ from diffusion?
Review Lab 1 Diffusion and Osmosis (U-tube) Not in dataloggers use this VDO to build your page in it. See instructor if you need assistance.
Data Collection Sheet
Quiz for Osmosis and Diffusion Lab
Review Lab 2 Osmosis (Dialysis)
Lab Procedure for Osmosis lab #2: This lab is in the dataloggers click the "Biology" file and then click on "Osmosis"
If you are unable to view the above or would like a printable copy please click on the button below.
Data Collection Sheet
Quiz for Osmosis Lab (dialysis bags)
Review Lab 3 Diffusion Intrusion This lab is not in the dataloggers so you will have to build a page.
If you are unable to view the above or would like a printable copy please click on the button below.
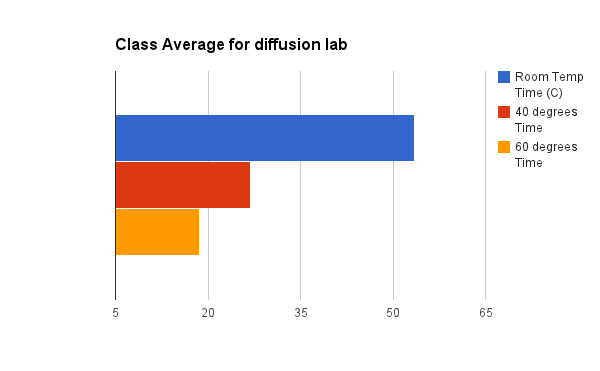
Below are class averages for each run. Notice that run 1 (room temperature) was the longest to record a +1 pH jump in 53.3 secs. Run 2 (40 degrees C) recorded a 26.9 seconds for the pH to increase by +1 pH point. The shortest time to increase by +1 pH point is Run 3 (60 degrees C) took 18.9 seconds.
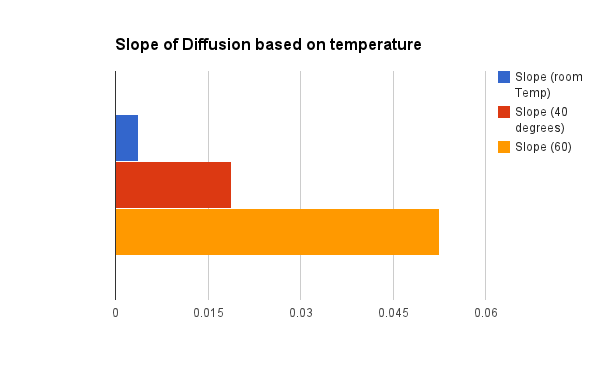
The graph below depicts how the increase in temperature affects the rate (how fast the particles are moving) of diffusion. In other words, the higher the temperature the faster the molecules are moving and the greater the likelihood particles will collide. The room temperature treatment had a .00387 slope , the 40 degree treatment's slope is .0188 and the 60 degree slope measured .0525. To put it another way the 60 degree treatment was traveling about 14 times faster than the particles in the room temperature group. So as the time of diffusion decreases the slope increases which creates more molecule collisions.