Atomic Models
The atom is the most basic unit of any element that still maintains the properties of that element. Because atoms are far too small to see, their structure has always been something of a mystery. For thousands of years, philosophers and scientists have proposed theories concerning the make-up of this mysterious particle, with increasing degrees of sophistication. Although there were many models, we will be studying two models, the Bohr's model and Lewis Dot Structures.
Level 11 Guiding Question: how can the periodic table be used to understand atoms?
Read the online chapter to find out to this question and how can it be used to draw atoms as well?
Level 11 Investigation #1: how can the periodic table be used to understand atoms?
Lewis Dot Structure
Answer the following three questions in your class comment section in your Google classroom:
- What is a single covalent bond?
- How can covalently-bound atoms obey the octet rule?
- Does the hydrogen molecule obey the octet rule?
Level 11 Investigation #2: how can the periodic table be used to understand atoms?
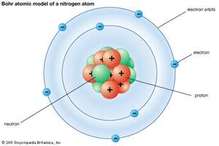
Bohr's Model
Click on the Blue button on the left to access the lesson on "How to make and understand what is the Bohr Model". Please read the short sections and complete each interactive activities (Structure of the Bohr Model, Bohr Model Practice, Drawing and Interpreting Bohr Models). To verify that you have completed the assignment please take a picture or screen capture each activity (see list the in the previous sentence) and up link in to your Google classroom. In addition there is a written assignment you will need to download using the grey button on the right.
Level 11 Videos: Learn how to draw and build the models
|
|
|
|
Level 11 Elaborate: film a lesson on how to drawing a Bohr and Lewis Dot model keep it up a minute.
Upload your video to your Google Classroom.